In 2004, Washington University in St. Louis researcher Jianmin Cui was handed a puzzling clue to the structure of an ion channel his lab had been studying for five years.
Scientists at the Cleveland Clinic Foundation in Ohio had located a large family whose members suffered from epilepsy, sudden attacks of involuntary movement or both, a syndrome called generalized epilepsy and paroxysmal dyskinesia (GEPD).
By analyzing the DNA of the 16 family members who had inherited the syndrome, the Cleveland Clinic scientists identified a mutation in the study family in the gene that encodes the big-conductance potassium ion channel (also known as the BK or MaxiK channel). This was one of the ion channels Cui, PhD, the Spencer T. Olin Associate Professor of Biomedical Engineering in the School of Engineering & Applied Science, had long studied.
Ion channels are small, highly selective pores in the cell membrane that allow specific ions (charged atoms) to enter or leave the cell. They are not continuously open but instead have gates, which open briefly and then close again in response to electrical, mechanical or chemical signals.
The mutation changes a single amino acid, or protein building block, in the BK channel.
The scientists thought that the mutation led to an increase in channel activity and thus nerve-cell excitability, causing epilepsy or involuntary movement, depending on which parts of the central nervous system were affected by the mutation.
But there was a problem. The altered amino acid was not part of the gate that opens and closes the channel.
Cui and his colleagues were thus faced with the problem of action at a distance. How did the mutation that apparently didn’t interfere with the channel’s gate nevertheless alter how often it was open?
Meticulous work that involved mutating other amino acids and testing the electrical properties of the mutated channels in frog eggs finally led to an answer, published in the June 24 issue of the journal Neuron.
It turns out that the mutation makes part of the channel more rigid, altering its dynamics in a way that makes it easier to toggle it open. This is the first time, says Cui, that protein dynamics have been implicated in the functioning of an ion channel.
Ion channels
Life itself depends on ion channels, the tiny pores in our cells that open on cue, letting charged atoms rush in or out. These influxes and effluxes create the electrical signals that make our eyes blink or our ribs expand to draw breath.
But because the channels are tiny and stuck in the greasy cell membrane, they are hard to study. Their existence wasn’t even confirmed until the 1970s. Working out the structure and function of a single ion channel (there are more than 300 types) earned researchers a Nobel Prize as recently as 2003.
Nerve impulses are actually changes in the electrical potential across a nerve cell’s membrane. The traveling wave of electrical excitation, known as an action potential, is orchestrated by the synchronized opening and closing of ion channels.
At rest, cells are slightly more negative (a matter of millivolts) inside than they are outside. An action potential starts when sodium channels open, allowing positively charged sodium atoms to rush into the cell, reversing the polarization of the membrane.
The sodium channels then automatically close and potassium channels open, repolarizing the membrane by allowing positively charged potassium ions to flood out of the cell.
What the BK channel does
One of the BK channel’s functions is to allow a nerve to adapt to constant stimulation by becoming less responsive. This loss of sensitivity is what allows us, for example, to ignore the constant pressure of our clothing.
Ion channels are gated, or triggered, in a variety of ways. Voltage-gated channels respond to the voltage, or potential difference, across the membrane. Ligand-gated channels are triggered by the binding of a chemical messenger, such as calcium. The BK channel’s gate responds to both types of stimuli.
Normally action potentials aren’t strong enough to trip the BK channel’s gate. But each action potential lets calcium ions into the cell. At higher calcium concentrations, the BK channel’s voltage threshold is shifted to lower voltages. Now action potentials can trip the gate, opening the BK channel.
The BK channel, also called the Big K channel or MaxiK channel, lets about 200,000 potassium ions through a millisecond, roughly 20 times more than a typical potassium channel. So when the BK channels open, they quickly repolarize the membrane, lopping off the action potential.
How might a BK channel mutation lead to epilepsy? Because the mutation allows the membrane to repolarize more quickly, the nerve cell can fire at higher frequencies, an excitability that may lead to seizures.
Cui emphasizes, however, that the link between the mutation and epilepsy has not been fully worked out.
The channel’s structure and mechanics
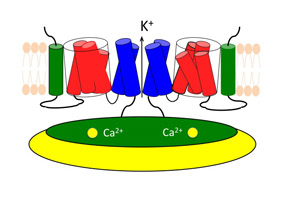
The various parts of the channel are only loosely associated, like chunks of pull-apart bread, and the voltage and calcium sensors are at some distance from the pore. So it is not at first clear how changes in the electrical field or the binding of calcium ions open the pore.
The voltage sensor includes a helix that is loaded with several copies of the positively charged amino acid arginine. The electric field across the membrane works on these positive charges, pushing the helix out of the membrane or pulling it deeper in, depending on the field’s polarity.
When it is pushed up, the helix pulls on the linker chain that connects the bottom of the voltage sensor to the bottom end of a helix in the pore. So pushing the charged helix up opens the pore, and pushing it down closes the pore.
What about the ligand gating? When calcium binds to the cystolic domain, it changes the cystolic domain’s shape (its conformation), and the domain tugs on another linker chain connected to one of the pore-lining helices as it warps.
The channel’s dynamics
In trying to clarify the effect of the mutation on BK channel function, Cui’s group uncovered yet another mechanism for action at a distance.
After the mutation’s discovery, experiments showed that it affects the pathway between a calcium-binding site on the cystolic domain called D367 and the channel’s pore. The epilepsy-causing mutation is very close to the binding site but not part of it.
So how was the mutation making it easier to open the channel?
Cui wondered if it didn’t have something to do with the mechanical properties of the amino acid that is substituted into the mutated form of the channel. The amino acid is glycine, which is exceptionally flexible; the presence of gylcine can allow a protein to bend at that position like a mechanical joint.
The scientists’ idea was that this local flexibility might actually make the pathway between the D367 binding site and the pore more rigid, which in turn might make the channel more sensitive to calcium binding.
To see whether dynamics mattered, Cui’s team immersed wild type channels in thick solutions of sugar or glycerol. The viscous solutions, which mimicked a more rigid D367 pathway, did indeed increase the channel’s sensitivity to calcium.
To explore further, Cui’s team then simulated the D367 pathway and its dynamics. The simulation showed that the wild-type protein is more flexible than the mutated protein and that glycine did indeed reduce the flexibility of other areas of the pathway.
The effect of the mutation, Cui concluded, was to make the D367 pathway move as a more rigid entity that couples the calcium binding site more tightly to the pore, so that it was easier to toggle the channel open and closed.
What it all means
As far as he knows, Cui says, this is the first study to focus on the dynamics of ion channels and to show that dynamics matter to function.
He suspects, however, that dynamics might be generally important to ion-channel function. A class of ion channels called TRP channels, Cui says, respond to heat or cold — and also to menthol, which is why menthol is a “cool” taste. At colder temperatures, proteins would move more sluggishly, so in this case, too, it is possible that dynamics influence function.
Understanding ion channels is also important to medicine. Many poorly understood diseases, such as epilepsy, migraine, cardiac arrhythmias and some autoimmune diseases, are being revealed to be disorders of the ion channels, or channelopathies, as they are called.
Based on what is known of the BK channel’s roles in the body, work on the channel might lead to the identification of several more channelopathies.
The BK channel is implicated not just in epilepsy and movement disorders but also in hypertension (it regulates the tone of smooth muscles like those that line the blood vessels). It is a suspect in schizophrenia because some antipsychotic drugs function much as BK does to increase outflows of potassium, and it may also play a role in autism and mental retardation.
In short, the basic research now underway in Cui’s lab may one day have an impact far beyond its walls.
***************************************
To learn more about the methods Cui uses to study ion channels, go to our video newsroom.