An innovative procedure that can replace a diseased heart valve is effective for patients who are too frail to endure open-heart surgery, according to results of a nationwide clinical study.
Doctors at Washington University School of Medicine in St. Louis and elsewhere have demonstrated that an investigational device, which uses a catheter to implant a uniquely designed replacement aortic valve, can make valve replacement an option for patients with aortic stenosis who are not suitable candidates for surgery.
The study involved older, critically ill patients who were not candidates for open-heart surgery, which requires stopping the heart and putting patients on a heart-lung bypass machine.
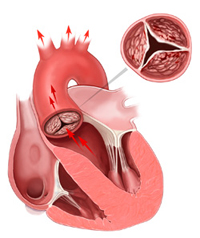
Instead, doctors used a catheter to thread the new valve through a leg artery into position within the heart while the patient’s heart continued beating. The technique is known as transcatheter aortic valve implantation.
The study appears online in the New England Journal of Medicine Sept. 22. Results of the randomized study were announced Sept. 23 at the Transcatheter Cardiovascular Conference in Washington, D.C. Edwards Lifesciences, maker of the valve, sponsored the trial.
The trial concluded that in patients with severe aortic stenosis who were not suitable candidates for surgery, transfemoral transcatheter aortic valve implantation was superior to standard therapy, markedly reducing the rate of death from any cause, the rate of death from cardiovascular causes and the rate of repeat hospitalization.
Edwards Lifesciences
The leaflets of a stenotic or calcified aortic heart valve are unable to open wide, obstructing blood flow from the left ventricle into the aorta. The narrowed valve allows less oxygenated blood to flow through and as a result, less oxygen-rich blood is pumped out to the body, which may cause symptoms like severe shortness of breath.
While major strokes were observed more frequently in the transcatheter aortic valve replacement group, the rate of the composite of major stroke or death from any cause was still significantly lower in that group than in the standard therapy group.
The cohort of the clinical trial, called the PARTNER trial (Placement of AoRTic traNscathetER valves), for which the data were released, enrolled 358 patients with severe aortic stenosis who were deemed unsuitable candidates for surgery by at least two surgeon investigators.
Patients too frail to undergo potentially life-saving valve replacement surgery face an extremely poor prognosis. Half do not survive more than two years after the onset of symptoms, such as shortness of breath, passing out and chest pain.
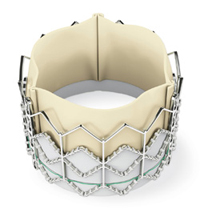
Fifty percent of the study patients had their diseased aortic valves replaced with the experimental valve, delivered via a catheter inserted through a small incision in the groin and navigated from the femoral artery into the heart, in much the same manner as a cardiac stent.
The other half was assigned to standard treatment for inoperable aortic stenosis, including medications and valvoplasty (balloon enlargement of the valve opening).
All patients were followed for at least one year. At the one-year follow-up, the rate of death from any cause was 30.7 percent in the transcatheter valve replacement group as compared with 50.7 percent in the standard-therapy group.
Washington University and Barnes-Jewish Hospital were among 21 heart centers (17 in the United States) selected to participate in this study.
Edwards Lifesciences
Edwards SAPIEN transcatheter heart valve
Ralph J. Damiano Jr., MD, chief of cardiac surgery at the School of Medicine, and Washington University interventional cardiologist John M. Lasala, MD, PhD, medical director of the cardiac catheterization laboratory at Barnes–Jewish Hospital, co-lead the St. Louis portion of the study. Co-investigators at Washington University include cardiac surgeon Hersh S. Maniar, MD, and interventional cardiologist Alan Zajarias, MD.
“The introduction of transcatheter valve replacement will go down as one of the innovative milestones in the history of cardiac surgery,” says Damiano. “It has the potential to dramatically reduce morbidity, particularly in elderly, high risk patients. We have seen a much quicker return to full activity in our patients with much less postoperative discomfort when compared to traditional open valve replacement through a sternotomy.”

Damiano
Lasala calls the device and technique a “game-changer” for many elderly with symptomatic aortic stenosis.“The incidence of aortic stenosis rises with age,” Lasala says. “Too often, however, age alone takes replacement surgery off the table as an option. But we have performed this procedure on patients up to age 94 with good outcomes.”
In an alternative procedure still under study, the experimental valve may also be inserted through an incision between the ribs (transapically) into the beating heart. This procedure is used for patients whose blood vessels are too small or contorted to accommodate the size of the catheter required.
Following closure of the randomized trial in March 2009, permission was granted by the FDA to offer the experimental procedure to any patient who qualifies due to moderate to severe risk of morbidity from traditional open-heart valve replacement.
Since 2008, 50 procedures using the technique have been performed by Washington University physicians at Barnes-Jewish Hospital, the only hospital in the region to use the procedure.
Patients who receive replacement aortic valves often feel their symptoms improve dramatically very soon after the procedure, the doctors say. Because the patients have not been subjected to the stress of invasive surgery, recovery time is shortened. Many patients report that they have resumed activities they have not enjoyed in years.
Lasala
Lasala says the new technology ranks with the development of medicated stents as one of the top advances in cardiology he has seen in his 23 years of practice.
A team, led by a surgeon and a cardiologist, is involved in all transcatheter aortic valve replacement cases. The name of the trial, PARTNER, signifies the important partnership between cardiac surgeons and interventional cardiologists (a specialist in catheter-based treatment of structural heart problems) who collaborate in valve patients’ evaluation, procedure and follow-up treatment, using a multi-disciplinary approach.
The complex procedure requires the integrated skill and collaboration of a cardiac surgeon, interventional cardiologist, echocardiographer and anesthetist. And, although it can be performed in a cardiac catheterization lab, a hybrid operating room, which enables immediate surgical intervention if needed, is the preferred location.
Zajarias traveled to France to learn the techniques from the inventor of the device, Alain Cribier, MD, head of cardiology at the University Hospital Charles Nicolle in Rouen, France.
“Dr. Cribier came to St. Louis to observe our first case, which took 2.5 hours,” Zajarias says. “Now we can do the procedure in under an hour, and we are seeing dramatic improvements in quality of life for our patients.”
The results of this particular trial cannot be extrapolated to other patients with aortic stenosis. A separate cohort of the PARTNER Trial is currently comparing transcatheter aortic valve implantation with surgical valve replacement among high-risk patients with aortic stenosis for whom surgery is a viable option and among low-risk patients with aortic stenosis. Those results are expected in 2011.
Leon M, et al. transcatheter aortic-valve implantation for aortic stenosis in patients who cannot undergo surgery. New England Journal of Medicine, Sept. 22 Online First.
More information about The PARTNER Trial (NCT00530894) may be obtained at www.clinicaltrials.gov.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.