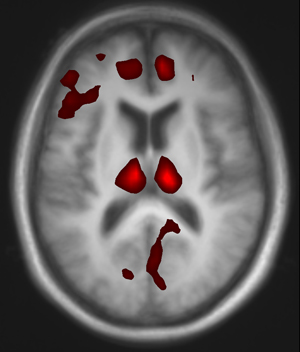
Neuroscientists using a new brain imaging technique could see an investigational drug for Parkinson’s disease get into a patient’s brain and affect blood flow in several key structures, an indicator the drug may be effective.
The study represents the first use of the technique in humans — called perfusion MRI — to test a drug still in development, the lead investigator says. In the future, similar brain scans could speed the development of new drugs and help clinicians learn whether established drugs are working, according to the researchers at Washington University School of Medicine in St. Louis.
The research team reports those findings Dec. 1 in The Journal of Neuroscience.
Perfusion MRI allowed the researchers to measure blood flow in the brain and even to determine the precise blood level of the drug, SYN115, needed to affect particular regions of the brain. They were able to demonstrate that the drug may be a viable therapy when combined with levodopa (L-Dopa), currently the most effective treatment for patients with Parkinson’s disease.
The study, a Phase 2 clinical trial, used two doses of the investigational drug. Its safety already had been tested in healthy volunteers. The goal was to determine what might be a reasonable dose for a larger clinical trial in Parkinson’s disease.
Unlike L-Dopa, which works through the brain’s dopamine system, SYN115 interacts with the brain’s adenosine A2a receptor. That receptor can modify the effects of the neurotransmitters glutamate and dopamine and is thought to be a potential therapeutic target not only for Parkinson’s disease but also for insomnia, pain, drug addiction and depression.

Black
“This study is important because it demonstrates this may be a useful approach for studying investigational drugs,” says lead investigator Kevin J. Black, MD. “We were able to determine both that the drug gets into the brain to exert its effects and that it has a larger effect at a higher dose.”
Black, a professor of psychiatry, of neurology, of neurobiology and of radiology, and his team used a new, FDA-approved type of perfusion MRI called arterial spin labeling (ASL), which uses MRI scans to measure blood flow in the brain. A similar technique uses positron emission tomography (PET) scans. Older forms of MRI scanning could not provide scientists with similar functional measurements.
Although PET scans can measure many of the same things, a large number of medical centers don’t have PET scanners, and depending how it’s done, PET may cost more, Black explains. The previous method of measuring blood flow with functional MRI is called BOLD scanning (Blood Oxygen Level Dependent).
“That’s a wonderful technique for watching someone’s brain respond to a question or a task or to compare blood flow changes over short periods of time,” he says. “But it’s not very helpful in determining the effects of drugs that might not influence the brain for 30 minutes or an hour. The ASL method works much better at comparing brain activity over that longer time period.”
SYN115 probably is not a candidate to replace L-Dopa, Black says, but if used with L-Dopa, he believes it may boost that drug’s therapeutic benefit and reduce its side effects. His team tested 21 Parkinson’s patients, comparing ASL functional MRI scans among patients who took only L-Dopa and those who took L-Dopa combined with either 20 milligrams or 60 milligrams of SYN115. Patients who received SYN115 experienced decreased blood flow in specific brain structures, with the biggest decreases occurring in the thalamus.
“The nerve cells that send signals to the thalamus are mostly inhibitory in nature,” Black says. “So we believe these decreases represent the brain taking its foot off of the brake pedal. The more drug in the system, the less ‘braking’ influence there is on the thalamus, which would then allow the thalamus to send positive signals to the brain’s cortex, where movements are initiated.”
Decreases in blood flow were more significant in subjects who had higher blood levels of the investigational drug. More study will be required to learn whether even higher doses might have a bigger influence on brain activity, Black says. It also will be important, he adds, to determine whether the decreased blood flow seen in the MRI scans will ease clinical symptoms of Parkinson’s disease, such as tremors, weakness, stiffness and difficulty walking.
But it is clear the drug is influencing brain function, he says. Even if this drug does not have a big impact, the method his team used to study the drug could influence future pharmaceutical research.
“This could shorten the time it takes to get medications to market because you don’t need as many patients or as much time to determine whether a drug has an effect in the brain,” he says. “This imaging technique could make it possible to significantly shorten the time from drug discovery to the launch of large, clinical studies to learn whether it truly is effective.”
And the technique has implications beyond Parkinson’s disease.
“One example might be depression,” Black says. “Antidepressant drugs work for some people and not for others. The drugs exert many effects within the first two hours, but often they don’t begin to relieve depression symptoms for a month or more. Theoretically, this imaging test could determine within hours whether we’re going to help patients, rather than waiting weeks to see if the drug is alleviating depression.”
Black KJ, Koller JM, Campbell MC, Gusnard DA, Bandak SI. Quantification of indirect pathway inhibition by the adenosine A2a antagonist SYN115 in Parkinson disease. The Journal of Neuroscience, vol. 30(48). pp. 16284-16292. Dec. 1, 2010.
This research was supported by a Synosia Therapeutics contract and by grants from the National Institutes of Health, the American Parkinson Disease Association’s Advanced Research Center for Parkinson Disease at Washington University and the Greater St. Louis Chapter of the American Parkinson Disease Association.
Kevin J. Black and Meghan C. Campbell performed personal consulting for Synosia Therapeutics, receiving payment of less than $10,000.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.