A key mechanism that appears to contribute to blood vessel damage in people with diabetes has been identified by researchers at Washington University School of Medicine in St. Louis.
Blood vessel problems are a common diabetes complication. Many of the nearly 26 million Americans with the disease face the prospect of amputations, heart attack, stroke and vision loss because of damaged vessels.
Reporting in the Journal of Biological Chemistry, the Washington University researchers say studies in mice show that the damage appears to involve two enzymes, fatty acid synthase (FAS) and nitric oxide synthase (NOS), that interact in the cells that line blood vessel walls.
“We already knew that in diabetes there’s a defect in the endothelial cells that line the blood vessels,” says first author Xiaochao Wei, PhD. “People with diabetes also have depressed levels of fatty acid synthase. But this is the first time we’ve been able to link those observations together.”

Wei is a postdoctoral research scholar in the lab of Clay F. Semenkovich, MD, the Herbert S. Gasser Professor of Medicine, professor of cell biology and physiology and chief of the Division of Endocrinology, Metabolism and Lipid Research.
Wei studied mice that had been genetically engineered to make FAS in all of their tissues except the endothelial cells that line blood vessels. These so-called FASTie mice experienced problems in the vessels that were similar to those seen in animals with diabetes.
“It turns out that there are strong parallels between the complete absence of FAS and the deficiencies in FAS induced by lack of insulin and by insulin resistance,” Semenkovich says.
Comparing FASTie mice to normal animals, as well as to mice with diabetes, Wei and Semenkovich determined that mice without FAS, and with low levels of FAS, could not make the substance that anchors nitric oxide synthase to the endothelial cells in blood vessels.
“We’ve known for many years that to have an effect, NOS has to be anchored to the wall of the vessel,” Semenkovich says. “Xiaochao discovered that fatty acid synthase preferentially makes a lipid that attaches to NOS, allowing it to hook to the cell membrane and to produce normal, healthy blood vessels.”
In the FASTie mice, blood vessels were leaky, and in cases when the vessel was injured, the mice were unable to generate new blood vessel growth.
The actual mechanism involved in binding NOS to the endothelial cells is called palmitoylation. Without FAS, the genetically engineered mice lose NOS palmitoylation and are unable to modify NOS so that it will interact with the endothelial cell membrane. That results in blood vessel problems.

“In animals that don’t have fatty acid synthase and normal nitric oxide synthase in endothelial cells, we saw a lot of leaky blood vessels,” Semenkovich explains. “The mice also were more susceptible to the consequences of infection, and they couldn’t repair damage that occurred — problems that also tend to be common in people with diabetes.”
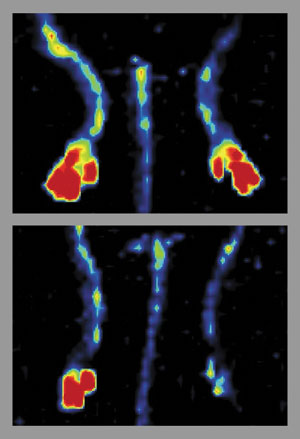
In one set of experiments, the researchers interrupted blood flow in the leg of a normal mouse and in a FASTie mouse.
“The control animals regained blood vessel formation promptly,” Semenkovich says, “but that did not happen in the animals that were modified to be missing fatty acid synthase.”
It’s a long way, however, from a mouse to a person, so the researchers next looked at human endothelial cells, and they found that a similar mechanism was at work.
“Our findings strongly suggest that if we can use a drug or another enzyme to promote fatty acid synthase activity, specifically in blood vessels, it might be helpful to patients with diabetes,” Wei says. “We also have been able to demonstrate that palmitoylation of nitric oxide synthase is impaired in diabetes, and if we can find a way to promote the palmitoylation of NOS, even independent of fatty acid synthase, it may be possible to treat some of the vascular complications of diabetes.”
And it shouldn’t matter whether a person has type 1 diabetes and can’t manufacture insulin or the more common type 2 diabetes, in which a person becomes resistant to insulin.
“That’s one of the key findings,” Semenkovich says. “It won’t matter whether it’s an absence of insulin or resistance to insulin: both are associated with defects in FAS.”
Wei X, Schneider JG, Shenouda SM, Lee A, Towler DA, Chakravarthy MV, Vita JA, Semenkovich CF. De novo lipogenesis maintains vascular homeostasis through endothelial nitric-oxide synthase (eNOS) palmitoylation, Journal of Biological Chemistry, vol. 286(4), pp. 2933-2945. Jan. 28, 2011.
This work was supported by grants from the National Institute of Diabetes, Digestive and Kidney Diseases and the National Heart, Lung, and Blood Institute of the National Institutes of Health and by awards from the American Heart Association and the American Diabetes Association.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.