Scientists have found that a protein made by a key Alzheimer’s gene slows the brain’s ability to get rid of amyloid beta, the main ingredient of the amyloid plaques that characterize the devastating illness.
The research, in humans and mice, links slow amyloid beta clearance rates to one form of the apolipoprotein E (APOE) gene.
The finding moves researchers closer to understanding a major risk factor for Alzheimer’s disease and may point to natural pathways for clearing amyloid beta that can be enhanced pharmaceutically. Scientists at Washington University School of Medicine in St. Louis report the results this week in Science Translational Medicine.
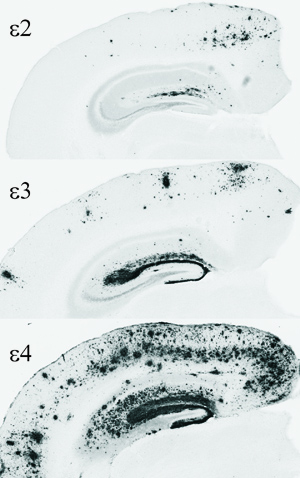
APOE comes in three forms: E2, E3 and E4. Neurologists have been interested because of genetic studies showing that APOE E4 substantially increases Alzheimer’s risk and lowers age of onset by 10 to 15 years.
“We knew that APOE was linked with amyloid beta accumulation and suspected that APOE E4 might slow amyloid-beta clearance. This study directly shows that this is particularly true for APOE E4,” says David Holtzman, MD, the Andrew B. and Gretchen P. Jones Professor and head of the Department of Neurology at Washington University. “The next step is to find out how APOE affects amyloid beta clearance and how APOE E4 disrupts that process, with the eventual goal of developing ways to enhance clearance.”
To begin the study, scientists analyzed the APOE genes in nearly 300 healthy human volunteers and used scans and cerebrospinal fluid to determine approximately how much amyloid plaque deposition the volunteers had in their brains. Those with one or two copies of the E4 form of the APOE gene were much more likely to have plaque deposition compared to individuals with other versions of APOE. Although all of the volunteers were healthy, amyloid plaque deposition and other brain changes associated with Alzheimer’s disease begin as much as 10 to 15 years or more before clinical symptoms become apparent.
APOE’s normal roles in the brain are still somewhat unclear. In the rest of the body, it circulates in the blood and plays an important role in controlling cholesterol levels.
Some causes of Alzheimer’s disease increase the brain’s production of amyloid beta, but scientists did not know whether APOE E4 caused more amyloid beta production or slowed its removal. To answer that question, Joseph Castellano, a doctoral student in Holtzman’s laboratory, worked with mice genetically altered to develop brain changes similar to Alzheimer’s and to exclusively make one of the three human forms of APOE.
Castellano used a technique called in vivo microdialysis to monitor amyloid beta levels in the brains of mice. He found that young adult mice that made APOE E4 had significantly higher levels of amyloid beta in their brains, and they cleared amyloid beta much more slowly than mice producing APOE E2 or E3. When he assessed amyloid plaque deposition in older mice, those that produced APOE E4 had many more plaques than mice that made APOE E2 or APOE E3.
To determine whether the different forms of APOE had any effect on amyloid beta production rates, Castellano applied another technique called stable isotope labeling kinetics in collaboration with the laboratory of Randall Bateman, MD, assistant professor of neurology at Washington University. The experiment showed no significant difference in production rates in mice with each of the three forms of human APOE.
“These experiments show that APOE E4 is impairing amyloid beta clearance compared to other forms of APOE,” Castellano says. “One very significant question is whether APOE and amyloid beta interact directly or indirectly. If they do bind to each other, does this binding differ according to the form of APOE, causing the differences in clearance we observed?”
Holtzman’s laboratory has already identified a receptor in the brain that removes APOE and amyloid beta.
“We would like to find out whether that receptor clears amyloid beta and APOE together, or if the two are removed from the brain through distinct mechanisms,” Holtzman says. “Answering these questions could be very important for new therapies.”
Castellano JM, Kim J, Stewart FR, Jiang H, DeMattos RB, Patterson BW, Fagan AM, Morris JC, Mawuenyega KG, Cruchaga C, Goate AM, Bales KR, Paul SM, Bateman RJ, Holtzman DM. Human apoE isoforms differentially regulate brain amyloid-beta peptide clearance. Science Translational Medicine, June 29, 2011.
Funding from the National Institutes of Health (NIH), the American Health Assistance Foundation, the NIH Neuroscience Blueprint Center Core Grant and Pfizer supported this research.
Washington University School of Medicine’s 2,100 employed and volunteer faculty physicians also are the medical staff of Barnes-Jewish and St. Louis Children’s hospitals. The School of Medicine is one of the leading medical research, teaching and patient care institutions in the nation, currently ranked fourth in the nation by U.S. News & World Report. Through its affiliations with Barnes-Jewish and St. Louis Children’s hospitals, the School of Medicine is linked to BJC HealthCare.