
Sometimes when people talk about solar energy, they tacitly assume that we’re stuck with some version of the silicon solar cell and its technical and cost limitations.
Not so.
The invention of the solar cell, in 1941, was inspired by a newfound understanding of semiconductors, materials that can use light energy to create mobile electrons — and ultimately an electrical current.
Silicon solar cells have almost nothing to do with the biological photosystems in tree leaves and pond scum that use light energy to push electrons across a membrane — and ultimately create sugars and other organic molecules.
At the time, nobody understood these complex assemblages of proteins and pigments well enough to exploit their secrets for the design of solar cells.
But things have changed.
At Washington University in St. Louis’ Photosynthetic Antenna Research Center (PARC) scientists are exploring native biological photosystems, building hybrids that combine natural and synthetic parts, and building fully synthetic analogs of natural systems, work that may one day lead to much-improved solar devices.
One team has just succeeded in making a crucial photosystem component — a light-harvesting antenna — from scratch. The new antenna is modeled on the chlorosome found in green bacteria.
Chlorosomes are giant assemblies of pigment molecules. Perhaps Nature’s most spectacular light-harvesting antennae, they allow green bacteria to photosynthesize even in the dim light in ocean deeps.
Dewey Holten, PhD, professor of chemistry in Arts & Sciences, ard collaborator Christine Kirmaier, PhD, research professor of chemistry, are part of a team that is trying to make synthetic chlorosomes. Holten and Kirmaier use ultra-fast laser spectroscopy and other analytic techniques to follow the rapid-fire energy transfers in photosynthesis.
The team’s latest results, described in a recent issue of New Journal of Chemistry, were highlighted in the editor’s blog.
Chlorosomes
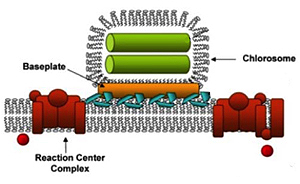
Blankenship/WUSTL
The photosystem in green bacteria consists of a light-harvesting antenna called a chlorosome and a reaction center. The energy of the light the pigments absorb is transferred to the reaction center (red) through a protein-pigment antenna complex called the baseplate (gold). The antenna (green) is made of rod-shaped aggregates of pigment molecules.
Biological systems that capture the energy in sunlight and convert it to the energy of chemical bonds come in many varieties, but they all have two basic parts: the light harvesting complexes, or antennae, and the reaction center complexes. The antennae consist of many pigment molecules that absorb photons and pass the excitation energy to the reaction centers.
In the reaction centers, the excitation energy sets off a chain of reactions that create ATP, a molecule often called the energy currency of the cell because the energy stored ATP powers most cellular work. Cellular organelles selectively break those bonds in ATP molecules when they need an energy hit for cellular work.
Green bacteria, which live in the lower layers of ponds, lakes and marine environments, and in the surface layers of sediments, have evolved large and efficient light-harvesting antennae very different from those found in plants bathing in sunlight on Earth’s surface.
The antennae consist of highly organized three-dimensional systems of as many as 250,000 pigment molecules that absorb light and funnel the light energy through a pigment/protein complex called a baseplate to a reaction center, where it triggers chemical reactions that ultimately produce ATP.
In plants and algae (and in the baseplate in the green bacteria) photo pigments are bound to protein scaffolds, which space and orient the pigment molecules in such a way that energy is efficiently transferred between them.
But chlorosomes don’t have a protein scaffold. Instead, the pigment molecules self -assemble into a structure that supports the rapid migration of excitation energy.
This is intriguing because it suggests chlorosome mimics might be easier to incorporate in the design of solar devices than biomimetics that are made of proteins as well as pigments.
Synthetic pigments
The goal of the work described in the latest journal article was to see whether synthesized pigment molecules could be induced to self-assemble. The process by which the pigments align and bond is not well understood.
“The structure of the pigment assemblies in chlorosomes is the subject of intense debate,” Holten says, “and there are several competing models for it.”
Given this uncertainty, the scientists wanted to study many variations of a pigment molecule to see what favored and what blocked assembly.
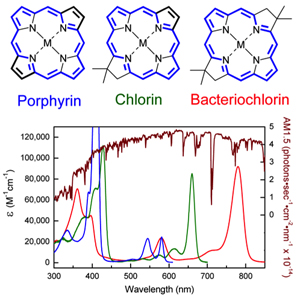
Holten/WUSTL
Nature provides three starting points for the design of synthetic pigments: porphyrin, chlorin and bacteriochlorin. Each of these macrocyles has an alternating double-bond pathway (in blue) that gives the molecule its basic electronic properties, including the ability to absorb visible or near infrared light. Hemoglobin is a porphyrin that lends blood its red color; chlorophyll, the pigment in green plants, is a chlorin; and the pigments in purple photosynthetic bacteria are bacteriochlorins. As the color-coded absorption spectra show, the three types of pigments absorb different colors of sunlight (brown).
A chemist wishing to design pigments that mimic those found in photosynthetic organisms first builds one of three molecular frameworks. All three are macrocycles, or giant rings: porphyrin, chlorin and bacteriochlorin.
“One of the members of our team, Jon Lindsey, can synthesize analogs of all three pigment types from scratch,” Holten says. (Lindsey, PhD, is Glaxo Professor of Chemistry at North Carolina State University.)
In the past, chemists making photo pigments have usually started with porphyrins, which are the easiest of the three types of macrocycles to synthesize. But Lindsey also has developed the means to synthesize chlorins, the basis for the pigments found in the chlorosomes of green bacteria. The chlorins push the absorption to the red end of the visible spectrum, an area of the spectrum scientists would like to be able to harvest for energy.
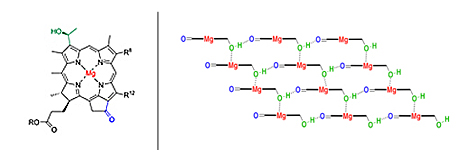
Key to pigment self-assembly are the metal atoms and hydroxyl (OH) and carbonyl (C=O) groups in the pigment molecules (the groups shown in color in the above illustration).
Doctoral student Olga Mass and co-workers in Lindsey’s lab synthesized 30 different chlorins, systematically adding or removing chemical groups thought to be important for self-assembly but also attaching peripheral chemical groups that take up space and might make it harder for the molecules to stack or that shift around the distributions of electrons so that the molecules might stack more easily.
Testing for aggregation
The powdered pigments were carefully packaged and shipped by Fed Ex (because the Post Office won’t ship chemicals) to Holten’s lab at WUSTL and to David Bocian’s lab at the University of California, Riverside.
Scientists in both labs made up green-tinctured solutions of each of the 30 molecules in small test tubes and then poked and prodded the solutions by means of analytical techniques to see whether the pigment had aggregated and, if so, how much had formed the assemblies.
Holten’s lab studied their absorption of light and their fluorescence (which indicated the presence of monomers, since assemblies don’t normally fluoresce), and Bocian’s lab studied their vibrational properties, which are determined by the network of bonds in the molecule or pigment aggregate as a whole.
In one crucial test Joseph Springer, a PhD student in Holten’s lab, compared the absorption spectrum of a pigment in a polar solvent that would prevent it from self-assembling with the spectrum of the pigment in a nonpolar solvent that would allow the molecules to interact with one another and form assemblies.
“You can see them aggregate,” Springer says. “A pigment that is totally in solution is clear but colored a brilliant green. When it aggregates, the solution becomes a duller green and you can see tiny flecks in the liquid.”
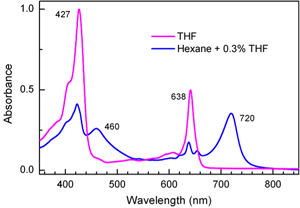
The absorption spectra indicated that some pigments formed extensive assemblies and that the steric and electronic properties of the molecules predicted the degree to which they would assemble.
Up next
Although this project focused on self-assembly, the PARC scientists have already taken the next step toward a practical solar device.
“With Pratim Biswas, PhD, the Lucy and Stanley Lopata Professor and chair of the Department of Energy, Environmental & Chemical Engineering, we’ve since demonstrated that we can get the pigments to self-assemble on surfaces, which is the next step in using them to design solar devices,” says Holten.
“We’re not trying to make a more efficient solar cell in the next six months,” Holten cautions. “Our goal instead is to develop fundamental understanding so that we can enable the next generation of more efficient solar-powered devices.”
Biomimicry hasn’t always worked. Engineers often point out early flying machines that attempted to mimic birds didn’t work and that flying machines stayed aloft only when nventors abandoned biological models and came up with their own designs.
But there is nothing predestined or inevitable about this. As biological knowledge has exploded in the past 50 years, mimicking nature has become a smarter strategy. Biomimetic or biohybrid designs already have solved significant engineering problems in other areas and promise to greatly improve the design of solar powered devices as well.
After all, nature has had billions of years to experiment with ways to harness the energy in sunlight for useful work.